Product Overview
Background
This kit allows you to measure total nitric oxide (NO) produced for in vitro experimental systems following conversion of nitrate to nitrite by metallic cadmium.
Nitric oxide can be spectrophotometrically assayed by measuring the accumulation of its stable degradation products, nitrate and nitrite. The ratio of these two products in biological fluids, tissue culture media, etc. may vary substantially. Hence, for accurate assessment of the total nitric oxide generated, one must monitor both nitrate and nitrite. An excellent solution to this problem involves the conversion of nitrate to nitrite catalyzed by cadmium. This is then followed by the quantitation of nitrite using the griess reaction.
This kit is suitable for the quantitative determination of total nitric oxide in samples that have high protein concentrations. The cadmium catalyst proves far more robust when in the presence of harsh deproteinating reagents than the alternative, nitrate reductase.
Assay Principle
In aqueous solution, nitric oxide rapidly degrades to nitrate and nitrite. Spectrophotometric quantitation of nitrite using Griess Reagent is straightforward, but does not measure nitrate. This kit employs metallic cadmium for quantitative conversion of nitrate to nitrite prior to quantitation of nitrate using Griess reagent — thus providing for accurate determination of total NO production.
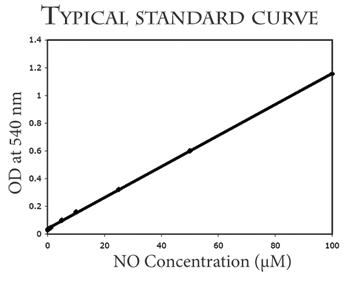
This kit can be used to accurately measure as little as 1 pmol/mL (~1mM) NO produced in aqueous solutions. Very little sample is required (1 to 100mL depending on the [NO] in the sample). The completed reaction is read at 540 nm.
References
-
Schmidt, H.H., et. al., (1995) Biochemica 2:22-23
